Slide Presenting the Life Sciences R&D Pipeline
RM0300025_9
- Last Update 11/07/2025
- File Size 1.6MB
- # of Slides 2
- File Format PPTX
- Slide Ratio 16:9
- Color
Keywords
- #Content-Based Slides
- #Design-Based Slides
- #Business Vision and Strategy
- #Technical Approach
- #Diagram
- #Business Strategy
- #Research and Development
- #Flow
- #Spread
- #Linear Flow
- #Centralized Diagram
- #16:9
- #R&D pipeline diagram PowerPoint
- #life sciences slide template
- #radial diagram PPT
- #research development process slide
- #clinical trial presentation
- #healthcare PowerPoint template
About the Product
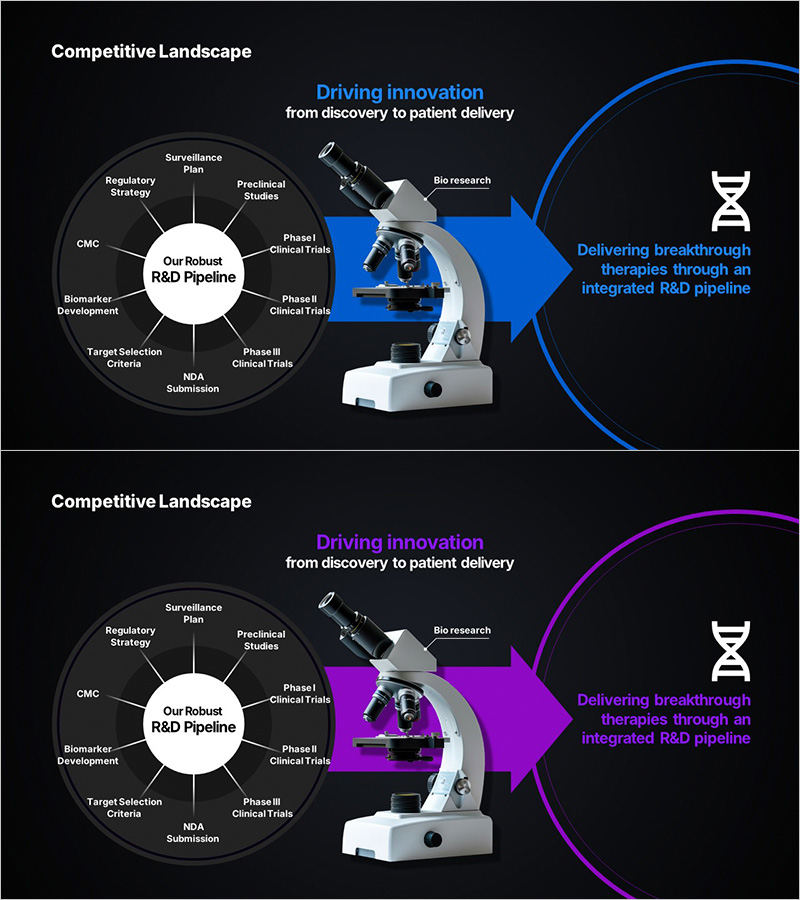
A PowerPoint slide that visualizes the research and development pipeline for life sciences and healthcare sectors. The diagram features a radial structure with the core R&D pipeline at the center, expanding outward to show all development stages from target selection through clinical trials to NDA submission. The slide is provided in two color versions—blue and purple—each highlighting different aspects of the development process. Key elements include target selection criteria, biomarker development, CMC, regulatory strategy, surveillance plan, preclinical studies, Phase I/II/III clinical trials, and NDA submission. The slide is ready for immediate use in presentations and allows full customization of text and shapes for your specific research pipeline.
Usage Points
-
Main Usage
Used to explain pharmaceutical and biotech companies' drug development processes to investors, employees, and partners. The radial structure emanating from the central R&D pipeline clearly illustrates each development phase—target selection, preclinical research, clinical trials, and regulatory approval—making complex processes intuitive and easy to understand.
-
How to Use
Insert into investor pitch decks, company overview presentations, or business plans in the technology and R&D sections. Choose the blue version to emphasize credibility and expertise, or the purple version to highlight innovation and differentiation. Customize each element's text to match your company's specific development pipeline and process.
-
Recommended For
Ideal for pharmaceutical and biotech company executives, investor relations managers, clinical trial coordinators, regulatory affairs specialists, and drug development project leaders. Particularly useful when presenting clinical trial results, regulatory submissions, or partnership proposals where clear explanation of development stages is critical.
-
Slide Structure
Two-slide set featuring a radial diagram with 12 elements radiating from a central hub: target selection criteria, NDA submission, Phase III clinical trials, Phase II clinical trials, Phase I clinical trials, preclinical studies, biomarker development, CMC, regulatory strategy, surveillance plan, preclinical research, and bio research. Includes microscope imagery and DNA logo to reinforce life sciences identity.
Related Products
-

Strategic Plan Keywords Diagram – Optimized for Business Strategy
#Business Introduction #Business Vision and Strategy #Diagram
-

Diagram Presenting Three Strategic Directions
#Business Vision and Strategy #Risk Management and Regulatory Compliance #Diagram
-

Key Maritime Strategy Diagram – Capturing Business Vision
#Business Vision and Strategy #Diagram #Business Vision
-

Central Diagram Slide for Cosmetic Manufacturing Strategy
#Business Vision and Strategy #Diagram #Business Vision


